Preparing for the FSMA Rollout With iAuditor
Checklist Best Practices | By | 20 Jul 2016 | 4 minute read

In order to strengthen the U.S. food safety program and protect public health, President Obama signed The FDA Food Safety Modernization Act (FSMA) on January 4, 2016. Previously the FDA primarily focused on reacting to food safety issues, with the new law in place the FDA is now able take proactive measures to prevent potential food safety problems.
Foodborne illnesses are a public health burden that is largely preventable. According to the Center for Disease Control and Prevention (CDC) each year roughly 1 in 6 Americans get sick, 128,000 are hospitalized and 3,000 die from foodborne illnesses. Enforcement of the FSMA will help reduce public health risk by overseeing and issuing guidelines on food recalls, imported food standards and domestic food standards in partnership with national food safety programs at state and local authority levels to ensure compliance.
SafetyCulture customers are using iAuditor to help them reach food safety compliance standards specified in new regulations of the FSMA. Conducting regular internal inspections with iAuditor can reduce exposure to unsafe food safety practices and promote production practices to prevent contamination and spread of foodborne illnesses.
How does the FSMA affect the Food & Beverage Industry?
Food Safety Risk Prevention
Under the FSMA, food facilities are required to implement a mandatory preventive controls plan that includes the following:
- Evaluation of hazards that have the potential to affect food safety
- Specify preventative controls that will be put in place to minimize or prevent the hazards
- Determine how the facility will monitor and ensure controls are working
- Maintain routine records of the monitoring
- Define actions that will be taken to correct problems
If your organization handles the production and harvesting of fruits and vegetables the guidelines stipulate further steps needed to identify naturally occurring hazards, introduced hazards (intentional or unintentional) and address soil amendments, hygiene, packaging temperature controls, and animals in the growing area and water. Additionally, the FDA will issue regulations to protect against the adulteration of food in order to protect the food supply chain at vulnerable points.
Using iAuditor, you can build food and food facility inspection checklists digitally or download an industry standard template from the SafetyCulture Public Library. Once the inspection template is complete, it is accessible from any mobile device loaded with iAuditor to visually
inspect cleanliness and sanitation of equipment and facilities as well as food handling habits of workers. iAuditor even allows you to build HACCP-based inspections to prevent food hazards during production. These practices are essential parts of Good Manufacturing Practices (GMPs).
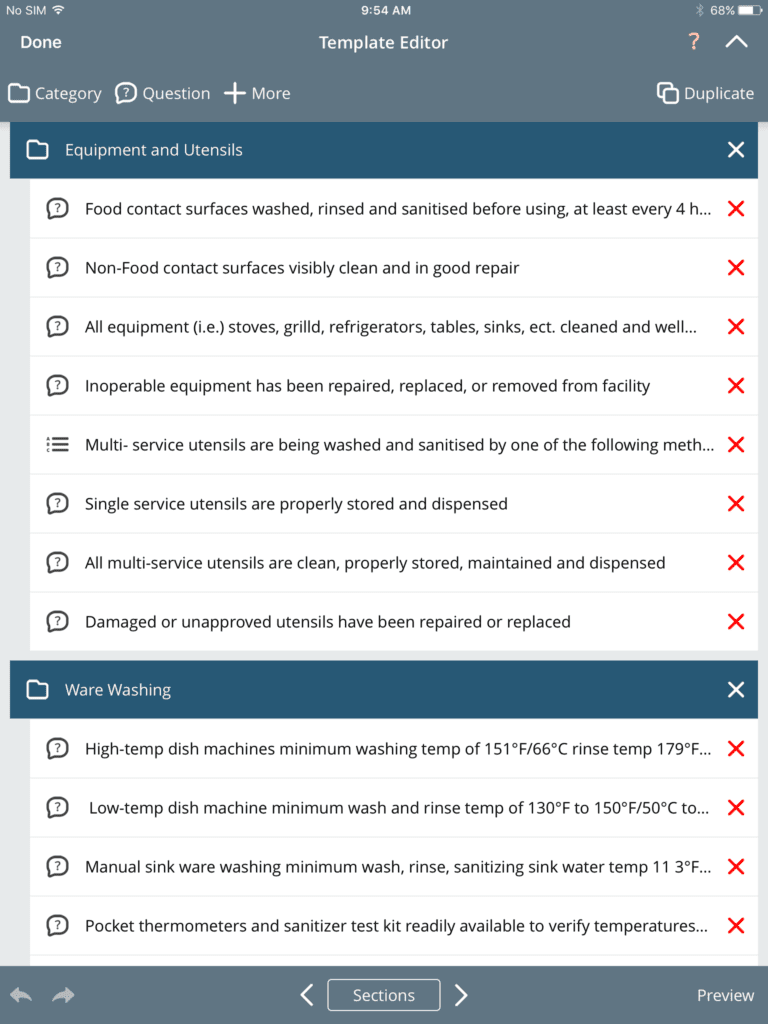 Example of food safety inspection template built in iAuditor
Example of food safety inspection template built in iAuditorFood Inspections and Compliance
The FDA will provide oversight to ensure compliance with new requirements of the FSMA and respond to any issues that arise. New guidelines and tools for inspection and compliance under the FSMA include the following:
- Immediate increase in the frequency of food inspections based on risk of the food facility. All high-risk domestic facilities must be inspected within five years and no less than every three years thereafter. The FDA will also inspect numerous foreign facilities every year for the next five years.
- Records firms will be required to document the implementation of their food safety plans allowing the FDA access to all records
- U.S. food testing laboratories must be FDA accredited laboratories that meet high-quality standards to carry out certain food testing
The FDA is reliant on inspections by other Federal, State and local agencies to meet the increased inspection mandate for domestic facilities. Over the next few years companies will need to perform internal audits more frequently in order to minimize overall food safety risks and have positive audit results with the increase of third party inspections. With iAuditor, food manufacturers are able to capture audit data digitally, take corrective actions to minimize food hazards, maintain records of all completed audits, and have analytics and real-time insight to ensure full compliance with FSMA.
Food Safety Response
If and when a food safety issue emerges outside of preventative controls, the FDA will have new authorities under the FSMA to respond effectively. These authorities include the following:
- Mandatory recalls for companies who fail to voluntarily recall unsafe food
- Flexible standard for detention of products that are potentially in violation of the law
- Registration suspension for a facility that poses a reasonable probability of adverse health consequences/death prohibiting them from distributing food
- Establish a track and trace system for both domestic and imported foods
- Issue a rulemaking requirement of recordkeeping for facilities that manufacture, process, pack or hold high-risk foods
Imported Foods
Food manufacturers are not the only companies affected by the FSMA, food importers will also need to maintain standards set forth in the new regulation. The FSMA impacts all imported products to ensure they meet U.S. standards and are safe for consumers. The following are new FDA authorities to protect imported foods:
- Explicit responsibility for importers to verify their foreign suppliers have adequate preventative food safety controls
- To facilitate imports, importers will now be required to acquire third party certification to certify that foreign food facilities comply with U.S. food safety standards
- High-risk imported foods are required to be accompanied by a credible third party certification or other assurance of compliance
- Establish a voluntary program for importers to expedite entry of foods for importers offering food from certified facilities.
- Refusal of entry from a foreign facility that denies FDA access to the facility or country which the facility is located
As regulations are increased, the need to facilitate imports becomes increasingly complex. Food importing companies will need to certify that they meet the new FSMA requirements. External audits performed by third party companies will highly impact a foreign food facility’s capability to conduct business in the U.S. If proper processes are put in place then are maintained and monitored using frequent internal auditing, food importers are able to increase their likelihood of acquiring accreditation.
Automated Internal Auditing Using iAuditor
Turning to digital solutions to keep pace with rapid regulation changes helps the food industry implement these new laws with much more ease. Most companies already have some form of auditing in place. By transforming the traditional paper-based method to an automated solution with a tool like iAuditor, you can conduct more food inspections faster and take immediate actions to mitigate any food safety risks. Maintaining high standards for hygiene, sanitary conditions, equipment and overall cleanliness in foodfacilities is essential to ensuring public safety from foodborne illnesses. To learn more about automating your food safety audits create a free iAuditor account.
Important Notice
The information contained in this article is general in nature and you should consider whether the information is appropriate to your specific needs. Legal and other matters referred to in this article are based on our interpretation of laws existing at the time and should not be relied on in place of professional advice. We are not responsible for the content of any site owned by a third party that may be linked to this article. SafetyCulture disclaims all liability (except for any liability which by law cannot be excluded) for any error, inaccuracy, or omission from the information contained in this article, any site linked to this article, and any loss or damage suffered by any person directly or indirectly through relying on this information.